High Flow Therapy* was provided for use in accordance with FDA’s guidance, “Enforcement Policy for Ventilators and Accessories and Other Respiratory Devices During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency,” Section IV Policy for Modifications to FDA-Cleared Devices, issued March 2020. The V60 Plus ventilator and high flow therapy upgrade, are not FDA cleared.
Philips Respironics is complying with the FDA’s Transition Plan for Medical Devices That Fall Within Enforcement Policies Issued During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency guidance document, by removing the high flow therapy (HFT or HFT60) software option from all V60 and V60 Plus devices in the US.
*High flow therapy is not for use on the V60/V60 Plus ventilators in the United States after November 7, 2023.
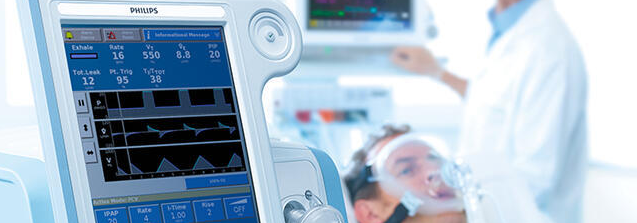
Welcome to the V60 and V60 Plus
Training Videos
Setting up HFT with SW 3.00 and 3.10
Transitioning from NIV to HFT with the AC611 HFNC on the Philips V60 Plus

Pocket Guides

Pocket Guide - V60 and V60 Plus ventilators
A step-by-step reference guide on how to use the ventilators

Pocket Guide - V60 ventilator
A step-by-step reference guide on how to use the ventilator
